A disease That Requires Medication for life?
Tremors, muscle stiffness, and slowed movements. Do you know what these symptoms experienced by Parkinson’s patients have in common?
It is the gradual death of dopamine-producing neurons in the brain. Dopamine is a neurotransmitter essential for motor control, and when it becomes insufficient, the body’s movements no longer function properly.
The problem is that most Parkinson’s patients must undergo lifelong drug treatment, and long-term use causes serious side effects. Levodopa (L-DOPA) is the core medication for Parkinson’s disease, but long-term use induces dyskinesia in more than 50% of patients (2019, Movement Disorders Journal). Dopamine agonists are associated with severe psychiatric side effects such as impulse-control disorders, hallucinations, and depression.
This raises a critical question: Is there a way to protect neurons at the cellular level instead of relying solely on medication?
Discovering Mitochondrial Damage in Parkinson’s Disease
Recently, the neuroscience field has been focusing on mitochondrial dysfunction as a fundamental cause of Parkinson’s disease.
Mitochondria are the power plants of the cell, the key organelles that generate ATP (energy). As we age, oxidative stress damages mitochondrial DNA, ATP production declines, and ultimately dopamine neurons die.
This means the problem is not simply a lack of dopamine; it is the answer to why neurons die.
What’s especially important is that multiple studies show mitochondrial complex I activity is reduced by 30–40% in the brains of Parkinson’s patients. This directly indicates a decline in energy-production capacity and creates an environment in which nerve cells struggle to survive.
Parkinson’s disease is not just a problem of “Dopamine Deficiency”
Many people think of Parkinson’s disease simply as ‘a condition caused by a lack of dopamine.’ That’s only partly true. While Parkinson’s manifests as dopamine deficiency, its root cause is the collapse of cellular energy metabolism.
- Excessive mitochondrial fission leads to cellular energy depletion.
- Reduced mitochondrial fusion prevents the cell from repairing damage.
The key is not merely supplementing dopamine with medication, but restoring and protecting the neurons themselves. Managing symptoms and addressing the cause are two very different things.
How Photobiomodulation (PBM) Helps Restore Mitochondrial Function

PBM is not simply shining light on the body. It is a treatment that restores the cell’s energy-production system.
In particular, PBM uses specific wavelengths to stimulate cytochrome-c oxidase inside mitochondria, increasing ATP production. This is a fundamental cellular repair process that medications cannot achieve.
Rebalancing Mitochondrial Fusion and Fission
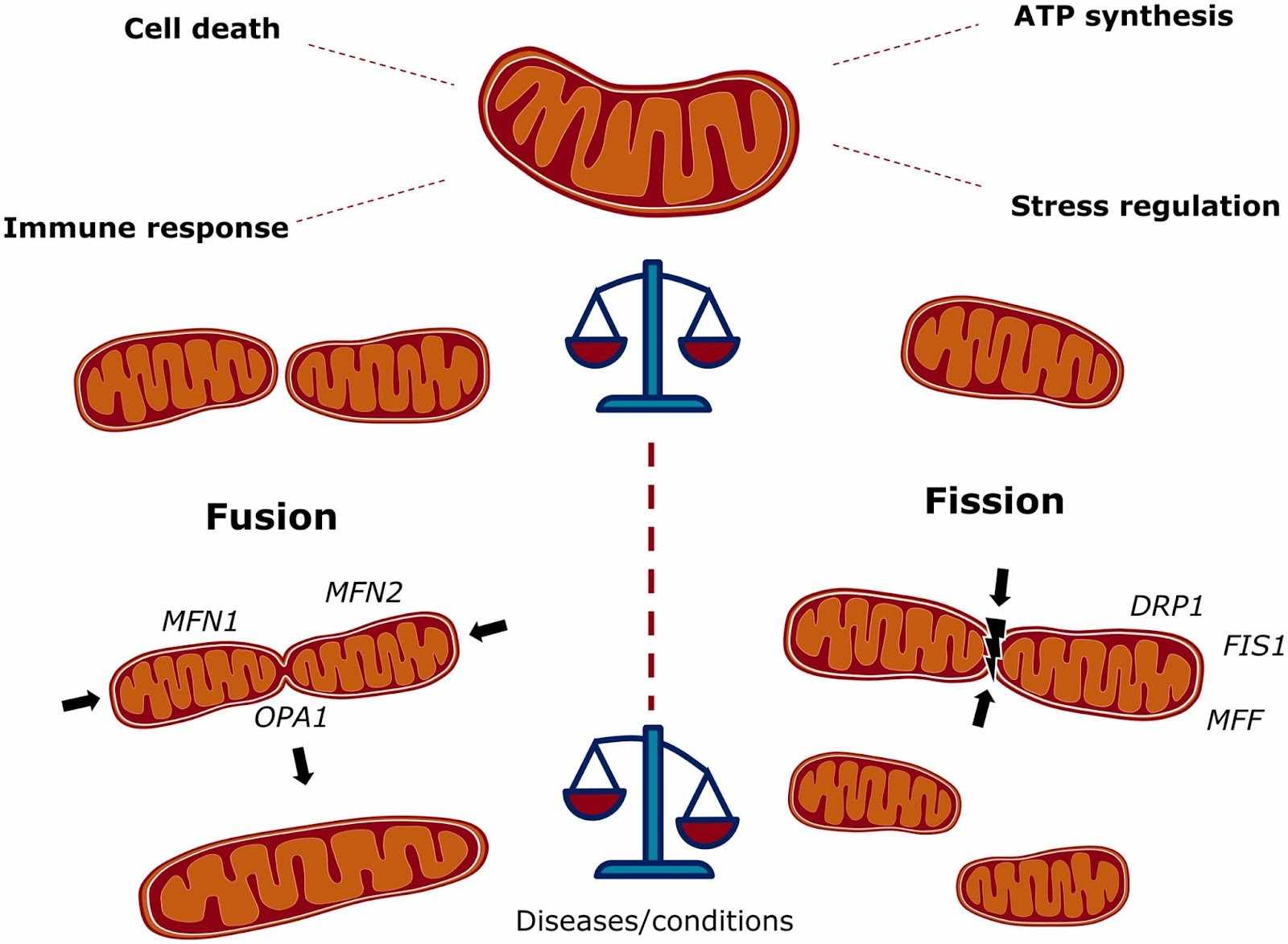
Mitochondria constantly repeat cycles of fusion and fission. When this balance collapses, cellular health collapses with it.
- Fusion proteins (MFN1, MFN2, OPA1): These merge mitochondria, repair damage, and improve energy efficiency.
- Fission proteins (DRP1, FIS1, MFF): These divide mitochondria, but when overactivated, they drive cellular energy depletion and cell death.
PBM reduces fission-inducing factors like DRP1 and FIS1, while increasing fusion-promoting factors like MFN1 and OPA1. Through this shift, mitochondria become healthier and more energy-efficient, preventing the cascade of dysfunction that leads to neurodegeneration (Trajano et al., 2024).
| Protein | Role | After PBM | Effect |
| DRP1 | Induces excessive mitochondrial fission (negative) | Decrease | Suppresses fragmentation → preserves energy and supports neuron survival |
| FIS1 | Supports DRP1-mediated fission (negative) | Decrease | Reduces cellular stress |
| MFN1 | Promotes outer-membrane fusion (positive) | Increase | Improves energy efficiency through larger, healthier mitochondria |
| OPA1 | Stabilizes the inner membrane and supports fusion (positive) | Increase | Maintains membrane potential and enhances ATP synthesis |
PBM also uses red light (660 nm) and near-infrared wavelengths (850 nm, 940 nm) that penetrate the skull and reach deep into brain tissue. This means PBM can act directly on neurons, unlike treatments that work only on skin or muscle.
Factors That Worsen Parkinson’s Disease
Research shows that the following elements impair mitochondrial function and accelerate Parkinson’s disease progression. Understanding these factors is critical for neuronal protection in Parkinson’s disease and strategies that support cellular energy restoration.
Oxidative Stress
Reactive oxygen species (ROS) directly damage mitochondrial DNA, proteins, and lipids, reducing ATP production capacity and making neurons more susceptible to degeneration. Chronic oxidative stress is a major driver of mitochondrial collapse in Parkinson’s patients.
Chronic Inflammation
Persistent neuroinflammation in the brain triggers immune responses that accelerate dopamine neuron death. Conditions like autoimmune disorders or systemic inflammation can exacerbate Parkinson’s progression. Anti-inflammatory interventions may support cellular energy restoration.
Environmental Toxins
Pesticides, herbicides, heavy metals, and air pollution inhibit mitochondrial complex I and disrupt energy metabolism. Prolonged exposure to such toxins increases oxidative stress and impairs neuronal survival. Reducing environmental toxin exposure can enhance mitochondrial resilience.
Lack of Sleep
Deep sleep is when the brain clears metabolic waste and repairs neuronal connections. Chronic sleep deprivation weakens neural recovery, reduces mitochondrial efficiency, and may accelerate cognitive decline in Parkinson’s patients.
Gut Microbiome Imbalance
Through the gut–brain axis, intestinal inflammation signals to the brain, affecting dopamine neuron health. More than 80% of Parkinson’s patients experience gastrointestinal symptoms such as constipation before motor symptoms appear. Maintaining a healthy gut microbiome may support neuronal protection and energy metabolism.
Physical Inactivity
Regular exercise promotes mitochondrial biogenesis, improves oxygen delivery to neurons, and enhances overall brain energy metabolism. Reduced movement lowers the brain cells’ ability to produce ATP, increasing vulnerability to neurodegeneration.
Poor Diet and Nutrient Deficiency
Deficiencies in key nutrients such as Coenzyme Q10, B-vitamins, magnesium, and antioxidants impair mitochondrial function. A nutrient-rich diet supports cellular energy restoration for neurodegeneration and helps maintain neuronal health.
Stress and Hormonal Imbalances
Chronic stress elevates cortisol levels, which can increase oxidative stress and inflammation in the brain. Hormonal imbalances, such as thyroid dysfunction, may also impair mitochondrial efficiency and worsen Parkinson’s symptoms.
Photobiomodulation therapy may help restore mitochondria damaged by these factors and improve the survival environment of nerve cells, supporting long-term neuron protection and energy restoration.
Clinical study: PBM improves walking speed in Parkinson’s Patients
For Parkinson’s patients, walking speed is not just a number. It directly reflects mobility, independence, and the risk of falling.
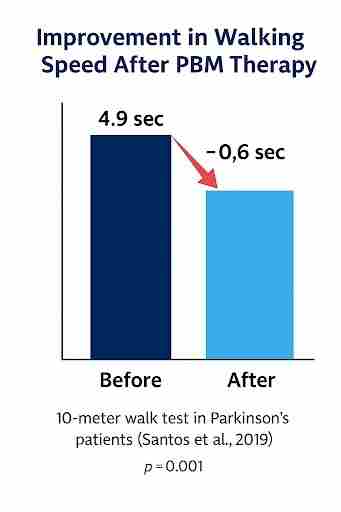
In a randomized controlled trial (RCT) published in Brain Stimulation by the Santos research team at the University of São Paulo in 2019, 35 Parkinson’s patients received photobiomodulation therapy for 9 weeks.
In the 10-meter walking test results:
The average walking speed improved by 0.6 seconds (from 4.9 seconds → 4.3 seconds, p = 0.001). This improvement exceeds the clinically meaningful minimum change (MCID, 0.23 m/s).
In other words, it represents a level of functional enhancement that patients can genuinely feel in daily life.
Another notable point from this study is that no side effects were reported. Unlike medication, PBM is non-invasive and does not harm the body.
The Future of Parkinson’s Treatment Lies Within the Cell
Approaching Parkinson’s disease as merely a matter of ‘suppressing symptoms’ has clear limits. The real goal is to keep neurons alive.
Photobiomodulation therapy does not artificially replace dopamine; it protects the neurons that produce dopamine. This mitochondria-restoring approach is supported by clinical research and has no known side effects. The era of relying solely on medication is fading. A therapy that protects neurons at the cellular level may be the breakthrough Parkinson’s patients have long awaited.
Your brain cells need energy right now. Shine light on your mitochondria. Movement returns.
Conclusion: Protecting Neurons Starts With Protecting Mitochondria
Parkinson’s disease begins long before symptoms appear at the cellular level, where mitochondrial damage starts. Restoring mitochondrial function offers a path forward that medication alone cannot provide.
PBM supports the cell’s energy systems, protects neurons, and may slow disease progression without harmful side effects.
For patients, this is not just treatment; it’s a chance to preserve independence, mobility, and quality of life.
Explore our medical-grade PBM devices today to protect mitochondrial health and support neuron survival.







